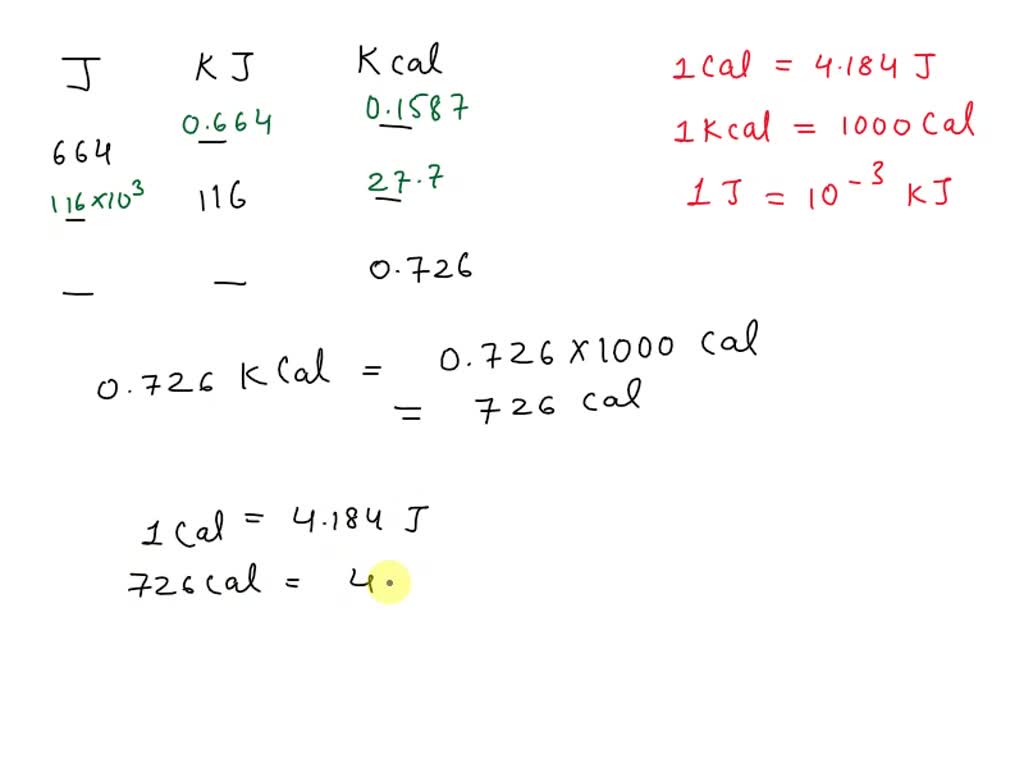
1 CH110 Chapter 2: Energy & Matter Potential & Kinetic Energy Energy from Food Temperature Specific Heat States of Matter Heating & Cooling Curves. - ppt download

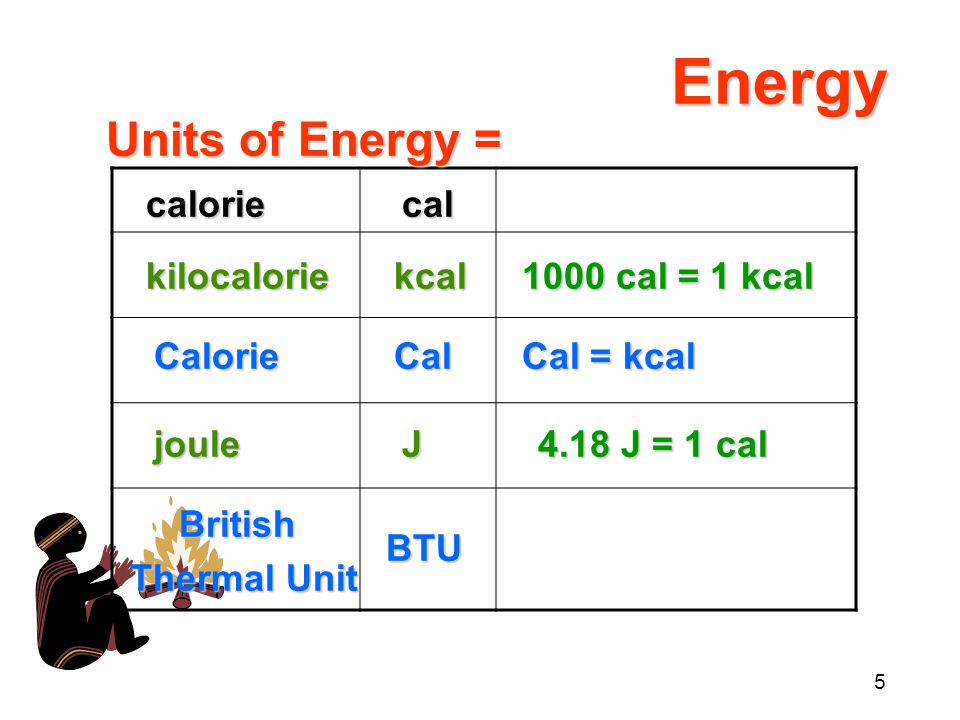
SOLVED: The Joule (J) is the SI unit of energy. 1 calorie (cal) = 4.184 J; 1 kcal = 1000 cal J kJ kcal 504 0.468 0.167